| History |
| Clinical symptoms |
| Vaccine and Treatment Updates |
|
|
| Home |
History |
In the late 1970's and early 1980's a new disease emerged in the United States and Europe that caused immunological disfunction in men who have sex with men. This was demonstrated by the rise of opportunistic infections such as Pneumocystis carinii pneumonia, Toxoplasma gondii encephalitis, and a number of unusual cancers like Kaposi Sarcoma in this population. In the months to come, this mysterious immunological deficency was found in several groups including hemopheliacs, blood transfusion recipients, haitian immigrants, and sexual partners or children of these groups. The distinct epidemiology of this suggested that the disease was transmitted through blood or sexual intercourse. In 1983, the Pasteur National Institute of Health discovered a virus molecule with a structure similar to the lymphadenopathy-associated retrovirus (LAV) discovered at the Pasteur Institute a few months back in one of these immunocompromised patients. The new retrovirus was classified as a member of the Lentivirusgenus and named Human Immunodeficiency Virus 1 . Later on, in 1986 another strain of the Human Immunodeficiency Virus was discovered (HIV-2) on the West coast of Africa. (1)
HIV-1 Versus HIV-2 The new strain of HIV (HIV-2) discovered off the West coast of Africa is distinctly different than the original HIV-1 strain. While they have almost the same set of genes and very similar pathological effects, HIV-2 bears greater resemblence to the Simian Immunodeficiency Virus (SIV). For example, while HIV-1, HIV-2 and SIV are all thought to share a common ancestor, both HIV-2 and SIV express an extra viral protein that HIV-1 does not. HIV-2 is also less pathogenic than HIV-1. (4)
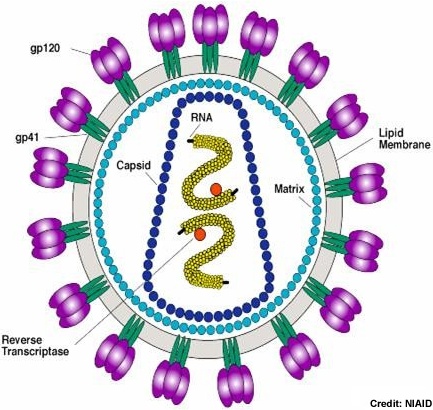
HIV-1 Virion Structure and Pathogenesis Virion Structure HIV-1 exhibits all of the structural characteristics of a typical lentivirus retroviridae. It was originally named a retrovirus because of its particle associated reverse transcriptase, a hallmark of the retroviridae family. It is a 100 to 120 µm enveloped virion with 2 single RNA strands and similar morphology to the lentvirus genus confirmed its retrovirus status.(1)
The HIV-1 virion uses the host cell membrane to form the viral envelope. This envelope is covered by gp41 and gp120 surface proteins i as well as Major histocompatability complex class II (MHC II) proteins inserted into the lipid envelope . Inside the lipid envelope, the matrix formed by Gag protein p17 holding the RNA containing core in place. The cylindrical core not only stores the viral RNA and various proteins, it also contains complementary RNA synthesized by the viral reverse transcriptase. (2)
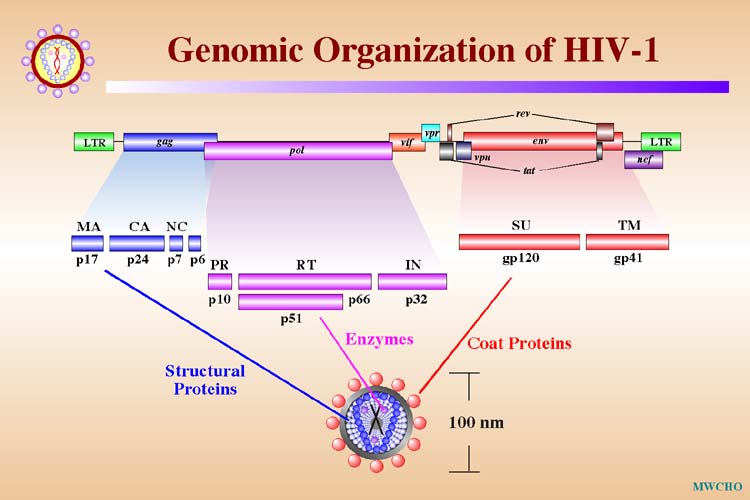
Genomic Organization
Transmission
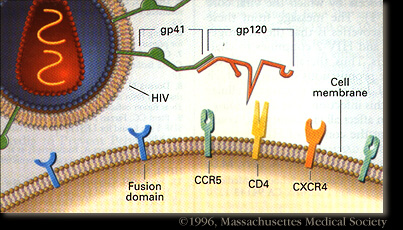
HIV-1 enters the blood stream most commonly through genital or colonic mucosa during sexual intercourse or other blood to blood contact. The virus enters the cell using the viral glycoprotein gp120 to interact with the host cellular recepter CD4 and chemockine receptor CCR5. The first cells targeted by the virus are usually the Langerhan cells, tissue dendritic cells of the lamina propia, and rarely direct infection of T cells. (2) The main cellular targets of HIV-1 are the CD4+ T-helper/inducer subset of lymphocytes, CD4+ cells of macrophage lineage and dendritic cells.
Clinical Symptoms Incubation The incubation period is about a few days to a few weeks after which individuals display acute- flu like symptoms. However after this short illness period, individuals can remain asymptomatic for years. (9) Symptoms For many people, early infection is asymptomatic. Within a month or two of exposure, one may display signs of fever, headache, fatigue, and enlarged lymph nodes. These symptoms usually fade for as little as a few months to up to 10 years depending on the individual. After this asymptomatic period, as the immune system is further broken down by the HIV virus more persistant symptoms such as energy and weight loss, fevers and sweats, persistent yeast infections, skin rashes or flaky skin, pelvic inflammatory disease, short term memory loss, and severe herpes infection appear.(3). Outcome There is no cure to HIV/AIDS. While there is very effective treatment called antiretroviral therapy, once someone has contracted HIV, they have it for life. Before treatment was developed, HIV patients would live between a few months to a few years after diagnosis as opportunistic infections quickly affected their immunocompromised state. With modern day therapy, that maintains a safe level of the HIV viral load, an HIV infected individual can live a normal adult lifespan but will have to take antiretrovirals every day of their lives. (3) Prevention and Management Antiretroviral therapy reduces the number of infected cells that allows an individual to preserve their immune response and lower the viral load in their bodies. Recent studies have boasted retrovirals as even showing long-term viral supression. (9). Highly Active Antiretroviral Therapy, a combination of three or more highly effective antiretroviral drugs are used to combat the HIV virus. It greatly reduces the viral load and by using combination therapy, reduces the rate of resistance against the drugs. The four main groups of drugs are Nucleoside Reverse Transcriptase Inhibitors, Non-Nucleoside Reverse Transcriptase Inhibitors, Protease Inhibitors, and fusion or entry inhibitors. (10) Drug Profile Lamivudine (3TC Nucleoside Reverse Transcriptase Inhibitor): 2',3'-dideoxy-3'-thiacytidine ( 3TC) Inhibits both HIV-1 and HIV-1 reverse transcriptase. It has also shown to be effective against hepatits B. It becomes activated after phosphorylated to its triphosphate state. Invented in Quebec in 1989 by Bernard Belleau and Nghe Nguyen-Ba and sold to Glaxo, a british pharmaceutical company. Molecular Information: Molecular Weight: 229.26 Emperic Formula: C8H11N3O3 Metabolism: Renal elimination (70%) Pregnancy category: C(USA) B3(Aus) Brand name: Epivir; Epivir-HBV Brand name of Combination products: Combivir (Lamivudine and Zidovudine) Epzicom(Lamivudine and Abacavir Sulfate) Why prescribed: Used in combination therapy twith zidovudine (Retrovir, AZT) o treat HIV infection. Commercial form: Tablet or liquid taken orally every 12 hours. Side effects : diarrhea, headache, fatigue, chills, upset stomach, vomiting, loss of appetite, dizziness, trouble sleeping, depression, stuffy nose, cough (11, 12, 13)
Vaccine and Treatment Updates Vaccine In May of 2004, the Walter Reed Army Institute of Research i n conjunction with the US National Institute of Allergy and Infectious disease began a US-based Phase I trial of LFn-p24, a new vaccine developed by Avant Immunotherapeutics. This novel vaccine combines an anthrax bacterium protein with the HIV Gag protein.(5) In June of 2004, researchers from Malawi and the US are working together to evaluate the vaccine candidate MRK-Ad5. In 2003 the US Center for Disease Control (CDC) is involved in the trials of two vaccine trials: VAX003 and VAX004 that are evaluating the gp 120-based vaccines produced by the private company VaxGen. In 2005, the CDC is working on developing new animal models to evaluate vaccines considering future vaccine trials in Africa and Thailand. (6) In 2002, the US NIAD worked with The Vaccine Research Center (VRC) on the Phase I clinical trials (VRC 004) of the multiclade, multigene DNA vaccine. This DNA vaccine uses the "prime-boost" strategy brought by the recombinant adenovirus type 5 (rAd5) acting as a vector to deliver HIV immunogens. This approach stimulates the immune system by first using a DNA priming vaccine and boosting later with the rAd5 vaccine. The initial study findings showed the vaccine was safe and effective. In December of 2003, a larger scale Phase I study was launched and in July of 2004, the first Phase I study of the booster vaccine was developed. In January of 2005, subjects of the original VRC 004 study received the booster vaccination to test the efficacy of the entire sequence. (7) To look at the most recent report on HIV Vaccine trials visit the latest IAVI report from 28 February 2005. http://www.iavireport.org/specials/OngoingTrialsofPreventiveHIVVaccines.pdf To visit the website of the AIDS vaccine 2006 Conference: http://www.aidsvaccine05.org/Home.aspx Antiretroviral Treatment (ARV )Treatment in Resource Limited Settings) The WHO's goal has been that by the end of 2005, 3 million people should have access to antiretroviral treatment. The WHO reccomends that in resource limited settings, HIV infected individuals should start ARV even if they are not showing clnical symptoms of AIDS. The first line regimin should consist of 2 nucleoside analogs and either a non-nucleoside or abacavir, or a protease inhibitor. Patients in these settings should have well documented past medical histories, current medical conditions, and other medication use. Since the WHO is just about to reach the 3 by 5 end term, an external evaluation of the initiative has been undertaken by a a Canadian International Development Agency. This final evaluation is set to come out on March 2006. They are set to be assessed on their accomplishments, lessons learnt, global leadership, alliances and advocacy initiatives taken. For more information on the WHO's 3 by 5 Initiative visit the WHO website.
References 2) Mandell, Gerald L. et al. Principles and Practice of Infectious Disease. Sixth Edition Churchill Livingstone. October 22, 2004. pp. 2122. 3) National Institute of Health. HIV Infection and AIDS: An Overview. US department of Health and Human Services. March 2005. <http://www.niaid.nih.gov/factsheets/hivinf.htm> 4) Lower human immunodeficiency virus (HIV) type 2 viral load reflects the difference in pathogenicity of HIV-1 and HIV-2 Popper SJ, Sarr AD, Travers KU, Gueye-Ndiaye A, Mboup S, Essex ME, Kanki PJ, Journal of Infectious Diseases, 180 (4): 1116-1121 OCT 1999. 5) International AIDS Vaccine Initiative. March 2001 IAVI Report. Nov. 14, 2005. <http://www.iavireport.org/VAX/VAXMay2004.asp>. 6) Center for Disease Control. CDC- HIV Vaccine Unit. Divisions of HIV/AIDS Prevention. Last Updated April 2005. <http://www.cdc.gov/hiv/vaccine/hivvu.htm>. 7) National Institute of Health. Vaccine Research Center: Scientific Updates. November 24, 2005. <http://www.niaid.nih.gov/vrc/scientificupdates_hiv.htm>. 8) Image Reference: UNAIDS Photo Archive. <http://www.unaids.org/bangkok2004/photos/C-2876_768.jpg>. 9)Altfield, Marcus and Bruce D. Walker. "Acute HIV-1 Infection." HIV Medicine. June 2005. <<http://www.hivmedicine.com/textbook/acuteinf.htm>. 10) Introduction ot HIV/AIDS November 2005. <www. avert.org> 11) Lamivudine. January 2003. <www.lamivudine.com >. 12) Medline Plus Lamivudine. November 2005. <http://www.nlm.nih.gov/medlineplus/druginfo/medmaster/a696011.html>. 13) Wikipedia. Lamivudine. <http://en.wikipedia.org/wiki/Lamivudine>. |
| Links! | ||
|
|
||
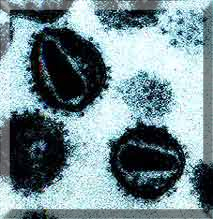 HIV-1 Virus. Reference: http://users.ox.ac.uk/~genemed/images/hiv1.jpg
HIV-1 Virus. Reference: http://users.ox.ac.uk/~genemed/images/hiv1.jpg HIV-2 Virus. Reference: http://www.csend.hu/magazin/0102/hiv2.jpg
HIV-2 Virus. Reference: http://www.csend.hu/magazin/0102/hiv2.jpg