
| Home | People | Publications | Funding | Journals | Links |
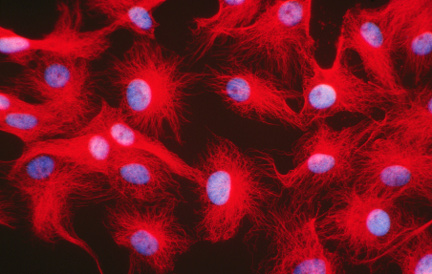
Microtubules (red) and nuclei (blue) revealed in cultured cells by fluorescence microscopy |
|
We study the organization and regulation of the microtubule cytoskeleton, and the relationship between the cell cycle and the cytoskeleton. Microtubules are polymers assembled from tubulin subunits, and are an essential element of the cytoskeleton. Microtubules are polar polymers, acting as directional tracks for the many motor proteins that move along them. Microtubules and motors move vesicles and organelles, and make up the spindle, which segregates chromosomes in mitosis and meiosis. How do cells organize microtubules into complex structures such as the mitotic spindle? One of the keys is the centrosome, which nucleates
microtubule polymerization from free tubulin subunits, and organizes the array of microtubules. We're interested in understanding the centrosome, and its role in cell division and differentiation.
We use the yeast Saccharomyces cerevisiae to study the genetics of microtubule function. The entire genome of this yeast has been sequenced, and the ease of genetic manipulation is unparalleled in any other eukaryote. Yeast cell biology isn't too bad either, despite the small size of yeast cells (check out some GFP-tubulin movies). We use eggs from the frog Xenopus laevis to study biochemistry of microtubule function. Frog eggs are very large cells from which it is easy to make concentrated cytoplasmic extracts that carry out many of the reactions of the intact egg. These reactions include centrosome formation on sperm, as occurs during fertilization, and centrosome duplication, as occurs each cell cycle in intact cells. Lastly, we use cultured cells from human, mouse and frog to study the cell biology of microtubule and centrosome function.
1) Cell cycle control of centrosome duplication. We have shown that duplication of the centrosome, the microtubule organizing center of animal cells, is dependent on the cell cycle kinase cdk2, and on cell cycle-specific proteolysis. We are now trying to determine the molecular mechanisms of centrosome duplication and to understand how centrosome duplication is controlled so that it happens once and only once per cell cycle. Cancer cells often have aberrant centrosome numbers, and we are investigating the relationship between aberrant centrosome number and the generation of cells with abnormal numbers of chromosomes.
2) Microtubule nucleation. Microtubules are polymers of tubulin, which is a heterodimer of alpha-tubulin and beta-tubulin. We have identified a remarkable complex of proteins associated with a third type of tubulin, gamma-tubulin. Gamma-tubulin and its associated proteins are localized to the centrosome and are critical for initiation, or nucleation, of microtubule assembly. The gamma-tubulin complex is a very large, ring-shaped complex and contains 5 proteins in addition to gamma-tubulin. We have now cloned all of these proteins, and are determining their role in microtubule nucleation.
3) Function of tubulin superfamily members. In addition to alpha, beta, and gamma-tubulin described above, we have identified two new tubulins in human cells, delta-tubulin and epsilon-tubulin. Both are localized to the centrosome, and we are working to determine their role in microtubule function. Epsilon-tubulin is asymmetrically distributed between the centrosomes after duplication; the old centrosome has epsilon-tubulin, whereas the new centrosome lacks it. Both delta-tubulin and epsilon-tubulin are conserved in vertebrates and some unicellular eukaryotes, but are absent from the sequenced genomes of fungi, plants, and lower animals.
4) Tubulin folding/biogenesis. Alpha-tubulin and beta-tubulin undergo a complex series of interactions with proteins termed tubulin cofactors. These interactions are thought to be involved in bringing together the two tubulins to make the tubulin heterodimer. The components of this pathway are conserved in all eukaryotes, and mutations in the yeast components cause defects in the microtubule cytoskeleton. We are interested in how these interactions help to make functional tubulin heterodimer, and whether they are involved in controlling the level of active tubulin in the cell.
|