

Background
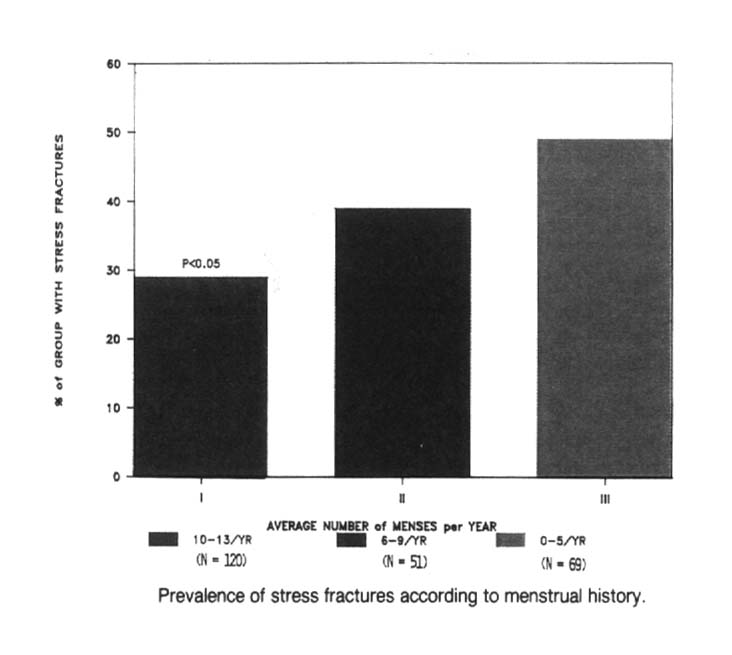
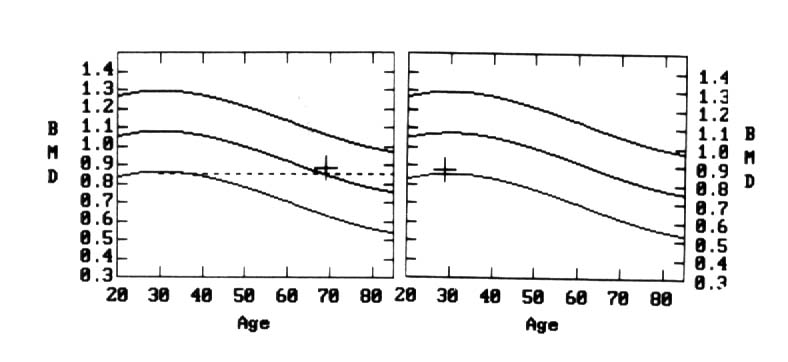
What are the Risks?

If you have questions, please contact:
Kristin Cobb, Project Director
or
Katrina Mogielnicki, Associate Director
Stanford University
Division of Epidemiology
Department of Health Research and Policy
HRP Redwood Building
Stanford, CA 94305
Phone: 650-498-6784
Fax: 650-725-6951
Toll Free: 1-877-RUN-BFIT
e-mail: kcobb@leland.stanford.edu
or katrinat@leland.stanford.edu
Study Investigative Team:
Jennifer Kelsey, Ph.D., Stanford University
Laura Bachrach, M.D., Stanford University
Robert Neer, M.D., Massachusettes General Hospital
MaryFran Sowers, Ph.D., University of Michigan
Gail Greendale, M.D., UCLA
Byron Brown, Ph.D., Stanford University
Kristin Cobb, Stanford University
Katrina Mogielnicki, Stanford University
This health study is made possible by funding from the
US Army Medical Research and Material Command.