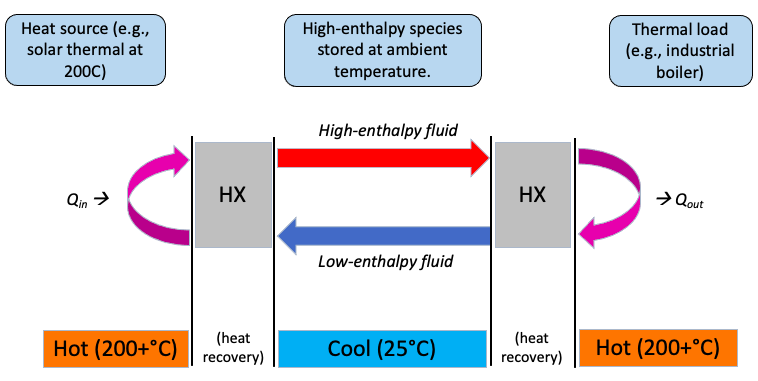
The final class in my department's graduate thermo series (ME370C) allows us to choose a project that applies the previous classes' tools for energy systems modeling. I decided to explore "chemical heat pipes," which I hoped to vet as a possible research topic. Most heat pipes, like those in computers, are "phase change heat pipes" in which a liquid evaporates at one end and condenses at the other end, rapidly moving heat from one side to the other. In principle, chemical bonds store more energy per unit of working medium than phase changes; chemical heat carriers could also be stored or transported at ambient temperature. I wondered whether reversible thermochemical reactions could store and transport heat better than the current best options (steam for heat transport and hot rocks or molten salt for heat storage) that are used in renewable thermal systems.
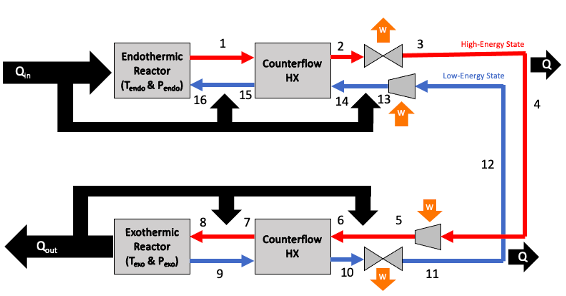
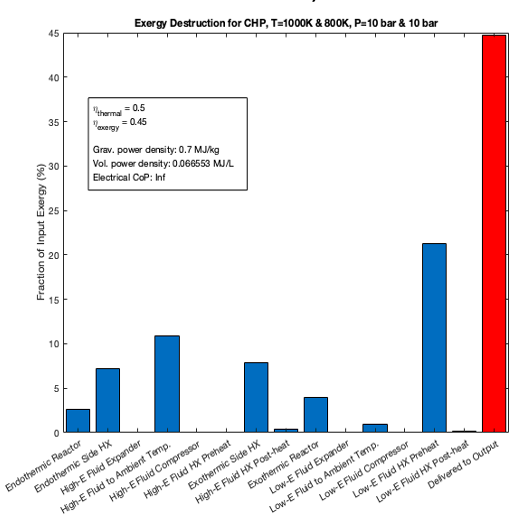
All of the coding for this project was done in MATLAB. The reactive and vapor-liquid equilibrium solvers were tested against results from Cantera. The model included an endothermic reactor at the heat source, an exothermic reactor at the heat sink, and pumps and counterflow heat exchangers for the working fluid. The working fluid circulates in a closed loop, with its composition swinging between high-enthalpy and low-enthalpy states. I was able to model first-law efficiencies, energy efficiencies, coefficients of performance, and power flow densities for arbitrary working fluids, reacting species, and operating temperatures and pressures.
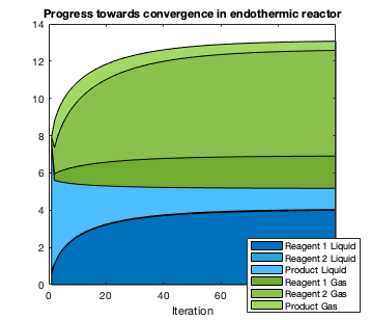
The most general version of the model required only the standard enthalpies, standard entropies, and boiling points of the reagents, all of which are tabulated even for obscure chemicals. Reactive vapor-liquid equilibria and heat fluxes could then be calculated at the temperatures of interest. Since the calculations became somewhat slow, I used MATLAB's parallel computing toolbox to split computations among multiple cores on my computer. I was able to model thermal storage using Diels-Alder reactions, a class of reaction that had seemed promising in literature but that used obscure organic reagents on which few data were available.
In the end, the chemical systems I modeled did not significantly outperform thermal storage and transport systems based on latent or sensible heats. Through conversations with other researchers, I learned that most of the substances I was considering had poor reaction kinetics or high viscosities, making them unlikely to work in real-world systems. Most importantly, the incumbent solutions (rocks, salt, or steam) use the cheapest possible materials, so the complexity and cost of a chemical system could not be justified given the modest performance gains expected. The project was an interesting and educational exercise, even though I chose not to pursue the research further.
(Click to return to project list)