 |
 |
Project Description
Purpose
Nonlinear finite element methods (FEM) have long been used to model deformations of soft tissue, but only recently has GPU computing evolved to the point where such methods can be performed in real time. The ability to model the dynamics of soft tissue in real time using FEM offers numerous benefits to both surgical simulation and planning. Currently, many real-time surgical simulations either use simplified mass-spring models or linear FEM, both of which suffer from inaccuracies under large strains or rotations. Nonlinear FEM would result in a more robust and realistic simulation. Furthermore, the incorporation of haptics imposes an additional computational cost, given that the force feedback loops in a realistic haptic simulation require frequencies on the order of 1 kHz, versus the 30-60 Hz used in graphics. Maintaining such high frequencies poses a significant computational challenge, especially when combined with the computational cost of nonlinear FEM. However, such a real-time platform would allow surgeons using virtual reality environments to save time in surgical planning without sacrificing prediction accuracy.
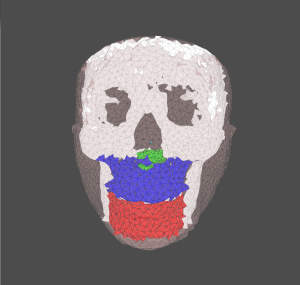
Although nonlinear real-time FEM has broad applicability, we illustrate the technique in the context of maxillomandibular advancement surgery, a clinical procedure used to reduce the severity of obstructive sleep apnea (OSA) and mitigate the risks of the co-morbidities associated with severe OSA. In this procedure, the surgeon wants to advance the maxilla and/or mandible enough to open the airway while minimizing undesired cosmetic alterations, e.g., prognathia. Since both the advancement and the airway diameter are typically measured in millimeters, surgical planning should be as accurate as possible.
Methods
Our specific FEM formulation currently uses a manually segmented tetrahedral finite element mesh with a St. Venant-Kirchoff constitutive model and Newmark time integration. We calculate the displacement at each time step using Newton-Raphson iteration. To assemble and solve the resulting linear systems, we intend to use OpenCL with existing algorithms to parallelize the computation on a GPU. Finally, we will implement 6-dof haptic interaction, in which the surgeon can use a Phantom Omni device to physically move a virtual fragment of the maxilla or mandible and feel the forces from the resulting soft-tissue displacements in real time. Our initial analysis will use standard material parameters for soft tissue and bone, but we are accumulating a database of pre- and post-surgery patient CT scans that will eventually enable empirical determination of material physical properties.
Results
We will present a real-time, haptic surgical planning environment to model changes in the diameter of the airway given a maxillomandibular advancement. This environment will rely on real-time nonlinear FEM on a GPU to simulate deformations in a soft-tissue mesh. Our evaluation of the software will include qualitative feedback from practicing oromaxillofacial surgeons, as well as numerical benchmarks for meshes of different sizes. For instance, we will be able to compare the predicted airway deformation with the actual post-operative change, using the normed difference in cross-sectional area of the airway integrated along the length of the throat. Based on previous results of GPU-based FEM, we expect to be able to model deformations in real time for meshes consisting of more than 10,000 tetrahedra.
Conclusion
By using parallel computing on a GPU for FEM, we can avoid the usual tradeoff between speed and accuracy to obtain a more realistic simulator for soft tissue. Having such a simulator operate in real time with a haptic interface would allow the surgeon to dynamically interact with a virtual environment in a realistic fashion. In particular, an oromaxillofacial surgeon would greatly benefit from a surgical planning tool that would model the airway volume changes possible with incremental advancements of the maxilla and/or mandible. The surgeon could then plan for the optimal increase in airway volume, given the challenge of balancing aesthetics with surgical feasibility.
On a more general level, the benefits of real-time nonlinear FEM extend to other clinical procedures. Any existing simulators that run slowly or provide rough linear approximations can improve performance or accuracy by using a GPU-based implementation of nonlinear FEM. Ultimately, improvements in the tools available for both surgical training and simulation lead to better clinical outcomes.
Project Staff
Status
Active since 2010.
Funding Sources
Funded through NIH Grant Number and the AO Foundation.