HIV Entry into Cells
HIV Membrane Fusion
To infect a cell, a virus must first be able to enter it. HIV is an enveloped virus and accomplishes cell entry by fusing the viral membrane with the cellular plasma membrane. This process is carried out by the viral envelope proteins gp120 and gp41. These two proteins are synthesized as a single 160 kD protein which is then cleaved. The products of this cleavage remain associated until the process of viral entry into the cell begins. gp120 binds to CD4 on CD4+ T lymphocytes and cells of the monocyte/macrophage lineage. This binding event and further interaction between gp120 and cellular co-receptors lead to gp120 dissociation from gp41. The dissociation of gp120 occurs as part of a conformational change in gp41 that leaves it in a "fusion-active" form. This form of gp41 can then mediate fusion between the cellular and viral membranes.
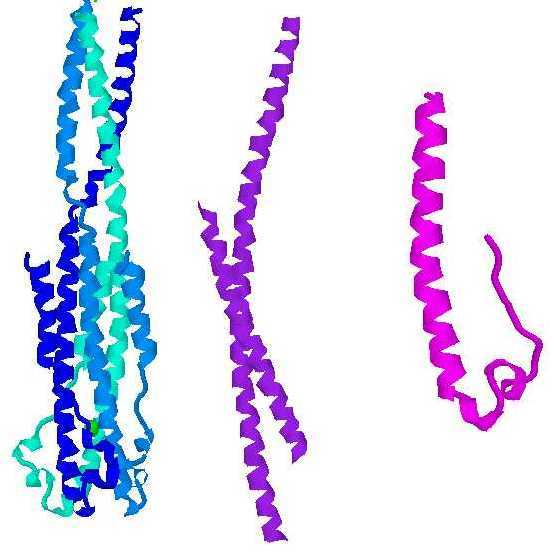
It is not known exactly how the process of HIV membrane fusion is accomplished. Insertion of a hydrophobic "fusion peptide" from gp41 into the target membrane is thought to bring the viral and cellular membranes into close proximity (Blacklow et al., 1995, by analogy from Yu et al., 1994). However, the process by which this apposition leads to fusion is unknown. Since a number of both viral and cellular proteins that carry out membrane fusion have similar structures, it is thought that the mechanisms of fusion may be similar in these different systems. The common structure is that of a helical coiled coil, in which three alpha helices wrap around each other. In the fusion-active state of many of these molecules, the helices undergo a reversal partway through the coiled coil and bend back on themselves, packing along the outside of the initial coil. (see image). This reversal places the transmembrane region and the fusogenic region near each other at one end of the coiled coil even though in the primary structure they are at opposite termini.

Images left to right: ectodomain of Ebola fusion protein, fragment of HIV gp41, portion of Moloney Murine Leukemia virus fusion protein (Weissenhorn et al., 1998; Weissenhorn et al., 1997; Fass et al., 1996). The HIV and MoMuLV proteins are found in the trimeric form seen with the Ebola protein. Images generated using Chemscape Chime from MDL.
Membrane Processes Leading to Fusion
Fusion is generally thought to occur via the formation of a pore between two closely apposed membranes. In its pre-fusion hairpin conformation, the fusion peptide is thought to be located in the target membrane, while the transmembrane region is in the viral membrane (as proposed in Weissenhorn et al., 1997 ; Chan et al., 1997). The coiled-coil structure forces these two membranes into close proximity. Subsequent to the formation of hairpin coiled-coil complexes, an unknown set of processes leads to fusion. In the post-fusion state, the hairpin conformation is retained, but both the fusogenic region and the transmembrane region are anchored in the same membrane (by analogy to models for influenza: Wharton et al., 1995; Bullough et al., 1994; Skehel et al., 1995). Based on analogy to the process of membrane fusion by influenza hemagglutinin, a process which has been extensively studied but is still not well-understood, it is likely that multiple hairpin structures interact in some way to form a narrow fusion pore in the apposed membranes, which then expands into a larger opening (Kanaseki et al., 1997; Danieli et al., 1996). Even if this analogy holds, the mechanism by which such a fusion pore is created is unknown and remains the subject of ongoing investigation.
Membrane Fusion as a Drug Target
One motivation behind the study of HIV fusion is the idea that an understanding of fusion events might allow one to block viral membrane fusion and thus prevent HIV from entering and infecting cells. One approach under investigation is the use of either peptides or small molecules to block formation of the fusion-active hairpin conformation by HIV gp41. Peptides derived from either the N-terminal or the C-terminal regions of gp41 can block viral entry (Wild et al., 1992; Wild et al., 1994; Jiang et al., 1993). gp41-derived peptides have also been used in clinical trials, and they appear to be effective in reducing viral load (Kilby et al., 1998). Approaches using small molecules to inhibit formation of the gp41 hairpin conformation are also under development. gp41 is a particularly attractive drug target because the amino acids comprising the area of gp41 to which hairpin inhibition drugs would bind is one of the most highly conserved sequences in HIV (Judice et al., 1997). A drug of this nature would be especially attractive because it stops HIV infection before cellular entry, reducing the likelihood that viral genetic material could be preserved inside the cell and lead to later reactivation.
References:
Blacklow SC, Lu M, Kim PS. A trimeric subdomain of the simian immunodeficiency virus envelope glycoprotein. Biochemistry 1995 Nov 21;34(46):14955-62.
Bullough PA, Hughson FM, Skehel JJ, Wiley DC. Structure of influenza haemagglutinin at the pH of membrane fusion. Nature 1994 Sep 1;371(6492):37-43.
Chan DC, Fass D, Berger JM, Kim PS. Core structure of gp41 from the HIV envelope glycoprotein. Cell 1997 Apr 18;89(2):263-73
Danieli T, Pelletier SL, Henis YI, White JM. Membrane Fusion Mediated by the Influenza Virus Hemagglutinin Requires the Concerted Action of at Least Three Hemagglutinin Trimers. J Cell Biology 1996; 133(3):559-69.
Fass D, Harrison SC, Kim PS. Retrovirus envelope domain at 1.7 angstrom resolution. Nat Struct Biol 1996 May;3(5):465-9.
Jiang S, Lin K, Strick N, Neurath AR. HIV-1 inhibition by a peptide. Nature 1993 Sep 9;365(6442):113.
Judice JK, Tom JYK, Huang W, Wrin T, Vennair J, Petropoulos CJ, McDowell RS. Inhibition of HIV type I infectivity by constrained alpha-helical peptides: Implications for the viral fusion mechanism. Proc Natl Acad Sci USA 1997;94:13426-13430.
Kilby JM, Hopkins S, Venetta TM, DiMassimo B, Cloud GA, Lee JY, Alldredge L, Hunter E, Lambert D, Bolognesi D, Matthews T, Johnson MR, Nowak MA, Shaw GM, Saag MS. Potent suppression of HIV-1 replication in humans by T-20, a peptide inhibitor of gp41-mediated virus entry. Nat Med 1998 Nov;4(11):1302-7.
Kanaseki T, Kawasaki K, Murata M, Ikeuchi Y, Ohnishi S. Structural Features of Membrane Fusion between Influenza Virus and Liposome as Revealed by Quick-Freezing Electron Microscopy. J Cell Biology 1997; 137(5):1041-1056.
Skehel JJ, Bizebard T, Bullough PA, Hughson FM, Knossow M, Steinhauer DA, Wharton SA, Wiley DC. Membrane fusion by influenza hemagglutinin. Cold Spring Harb Symp Quant Biol 1995;60:573-80
Weissenhorn W, Carfi A, Lee KH, Skehel JJ, Wiley DC. Crystal structure of the Ebola virus membrane fusion subunit, GP2, from the envelope glycoprotein ectodomain. Mol Cell 1998 Nov;2(5):605-16.
Weissenhorn W, Dessen A, Harrison SC, Skehel JJ, Wiley DC. Atomic structure of the ectodomain from HIV-1 gp41. Nature 1997 May 22;387(6631):426-30.
Wharton SA, Calder LJ, Ruigrok RW, Skehel JJ, Steinhauer DA, Wiley DC. Electron microscopy of antibody complexes of influenza virus haemagglutinin in the fusion pH conformation. EMBO J 1995 Jan 16;14(2):240-6
Wild C, Oas T, McDanal C, Bolognesi D, Matthews T. A synthetic peptide inhibitor of human immunodeficiency virus replication: correlation between solution structure and viral inhibition. Proc Natl Acad Sci U S A 1992 Nov 1;89(21):10537-41.
Wild CT, Shugars DC, Greenwell TK, McDanal CB, Matthews TJ. Peptides corresponding to a predictive alpha-helical domain of human immunodeficiency virus type 1 gp41 are potent inhibitors of virus infection. Proc Natl Acad Sci U S A 1994 Oct 11;91(21):9770-4.
Yu YG, King DS, Shin Y-K. Insertion of a Coiled-Coil Peptide from Influenza Virus Hemagglutinin into Membranes. Science 266, 274 (1994).
Unless otherwise noted, all contents are Copyright © 1999 by Peter Kasson.