Cilia-driven epithelial folding and unfolding in an early diverging animal
Abstract
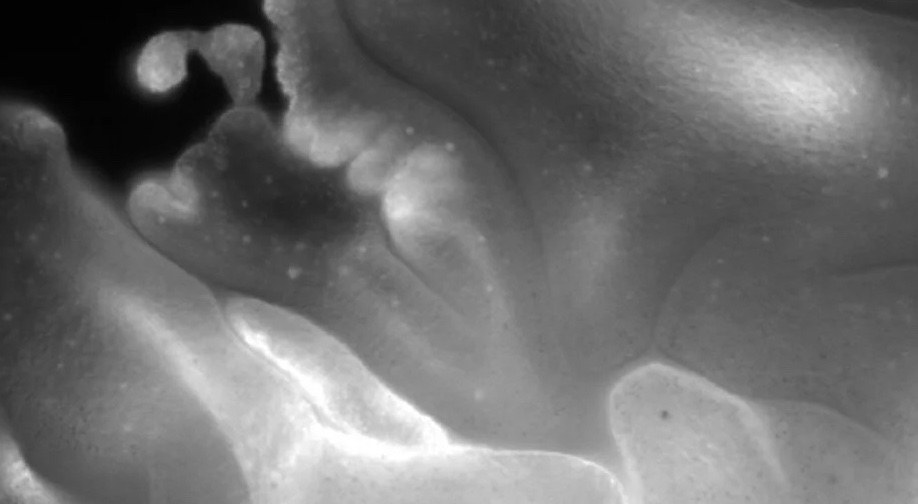
Multicellular organisms utilize epithelial folding to achieve remarkable three-dimensional forms. During embryonic development, stereotypical epithelial folds emerge from underlying active cellular and molecular processes including cell shape change and differential cell growth. However, the origin of epithelial folding in early animals and how folding may be harnessed in synthetic systems remain open questions. Here, we identify a modality of behavior-induced epithelial folding and unfolding arising from cilia–substrate adhesion and ciliary walking in the basal animal Trichoplax adhaerens (phylum Placozoa). We show that T. adhaerens is capable of exhibiting dynamic nonstereotyped folding states, providing a 3D perspective to an organism previously only characterized in its 2D state. We correlate these folding states to local substrate geometry, revealing that the animal conforms to available substrate surface area, promoting the maintenance of a folded state. Using 4D fluorescence light sheet microscopy, we characterize fold geometry, curvature evolution during unfolding, and the nonstereotypy of unfolding behavior. Through repeated unfolding trials, we reveal the robustness and timescales associated with unfolding behavior and employ scaling analysis and toy model simulations to establish how collective ciliary activity can robustly drive unfolding. In this way, despite lacking any folding–unfolding “pathway,” transitions between folding and unfolding states emerge as a function of the animal’s environment and motility. Our work reveals a remarkable behavior exhibited by a brainless, nerveless animal, and demonstrates the capacity for 3D–2D transitions in folding epithelial sheets using ciliary activity.