BIOLOGICAL CONCENTRATION REPORTERS:
Exploring a Dual GFP Construct at the Single Molecule Level

Figure 1: Schematic of the "Cameleon YC
2.1" construct and the principle of calcium concentration
detection by FRET described in the text. |
(Brasselet, Peterman, Miyawaki, and Moerner, J. Phys. Chem.
B (published on the web, March 2000), also J. Phys. Chem. B 104,
3676-3682 (2000))
One thrust of our research concerns the photophysical behavior
of single molecules of the Green Fluorescent Protein (GFP) and
its mutants. As is well-known, in cell biology and biochemistry,
GFP is currently widely used as an indicator for gene expression
or as a fluorescent label for a large variety of proteins. In
previous work, we observed single copies of GFP for the first
time (Dickson, Cubitt, Tsien, and Moerner, Nature, 355, 388 (1997)),
and our paper characterizing the fascinating single-molecule
blinking dynamics as a function of pH, host, mutant, and excitation
intensity has recently appeared (Peterman, Brasselet, and Moerner,
J. Phys. Chem. A 103, 10553 (1999)).
As a critical extension of this work, we have explored the single-molecule
dynamics of dual-GFP constructs designed to sense local ion concentrations
in biological media. A first study, recently completed, concerns
the "cameleon YC2.1" complex, whose structure is based
on a cyan-emitting GFP (CFP) separated from a yellow-emitting
GFP (YFP) by the calmodulin Ca2+-binding protein (CaM) and a
calmodulin-binding peptide (M13) (see Fig 1). This complex was
provided by R. Y. Tsien, and it was designed to allow sensing
of calcium ion concentrations in cells by fluorescence resonant
energy transfer (FRET). If Ca2+ ions are bound, CaM wraps around
M13, and the construct forms a more compact shape, leading to
a higher efficiency of excitation transfer from the donor CFP
to the acceptor YFP. The degree of FRET in cameleon is therefore
a sensitive ratiometric reporter of the concentration of Ca2+
in solution and cells. We measure FRET at the single-molecule
level using an ultrasensitive two-color confocal scanning microscope.
Analysis of single-molecule signals from the cameleon YC2.1
complex diluted in aqueous agarose gels allowed retrieval of
several interesting features of the energy transfer between the
donor and acceptor mutants of the construct, as a function of
the calcium concentration in the medium. The energy transfer
efficiency distribution deduced from single-molecule fluorescence
signals shows an increased width at the Ca2+ dissociation
constant concentration (see Figure 2). This observation is consistent
with the ligand binding kinetics, whose time scale at intermediate
calcium concentration is close to our measurement time scale
(20 ms). The complex dynamics of the fluctuations were examined
using a combination of autocorrelation and cross-correlation
in conjunction with polarization measurements at the bulk and
single-molecule level. Beside the reorientation fluctuations
of the two dipoles that seem to occur on a time scale fast compared
to our integration time (<20ms) but slow compared to the fluorescence
lifetime, we detected variations in the energy transfer between
the two GFP mutants on the 20-100 ms time scale. We found both
negative and positive cross-correlations in the donor and acceptor
emission signals, the former related to the energy transfer process,
and the latter presumably caused by other perturbations of the
donor and acceptor emission. A further conclusion of this work
is that single copies of this dual-GFP construct cannot accurately
report local calcium concentrations, chiefly due to the small
number of photons emitted before the termination of the on-time.
Future studies should explore methods to terminate the dark period
by secondary optical excitation.

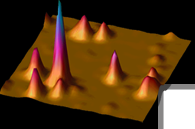
Figure 2: Histograms of the energy transfer efficiency
of cameleon YC2.1 in agarose deduced from single-copy measurements
of for three different concentrations of calcium: at
Ca2+ saturation, at an intermediate concentration (~ Kd),
and at very low calcium. |
|