Sherlock Lab Research: Evolution, using Yeast as a Model System
We have various interests (see publications for a more comprehensive idea
as to our research interests), but the lab's main focus is on the process
of evolution. We are interested in defining and understanding
evolutionary phenomena, both at the level of population dynamics, as well
as at the molecular level. We are interested in the following questions:
What is the rate of adaptive mutation? What is the distribution of
fitness effects for adaptive mutations? What are the identities of
adaptive mutations? Does the spectrum of adaptive mutations differ
between haploids and diploids? Do mutations that provide an adaptive
advantage under one condition provide an advantage under another? Or a
disadvantage (antagonistic pleiotropy). What is the underlying nature of
clonal interference? We are also interested in how the answers to these
questions can change as a function of genotype and environment. To answer
these questions, we use experimental evolution, and the budding
yeast, S. cerevisiae, as a model organism.
Our initial experiments in this area were using yeast grown in continuous
culture in a chemostat, with glucose as the limiting nutrient. Under
these conditions, we expected beneficial mutations to be selected, and to
increase in frequency. The fundamental question we sought to answer in
these early experiments was how pervasive is clonal interference. To
investigate this, we marked 3 otherwise identical subpopulations with
green, red and yellow fluorescent proteins, and then followed the sizes of
the subpopulations. We expected, based on the landmark Paquin and Adams
paper in 1983, that we would observe clear adaptive sweeps. Instead, we
saw pervasive clonal interference (Kao and Sherlock, 2008).
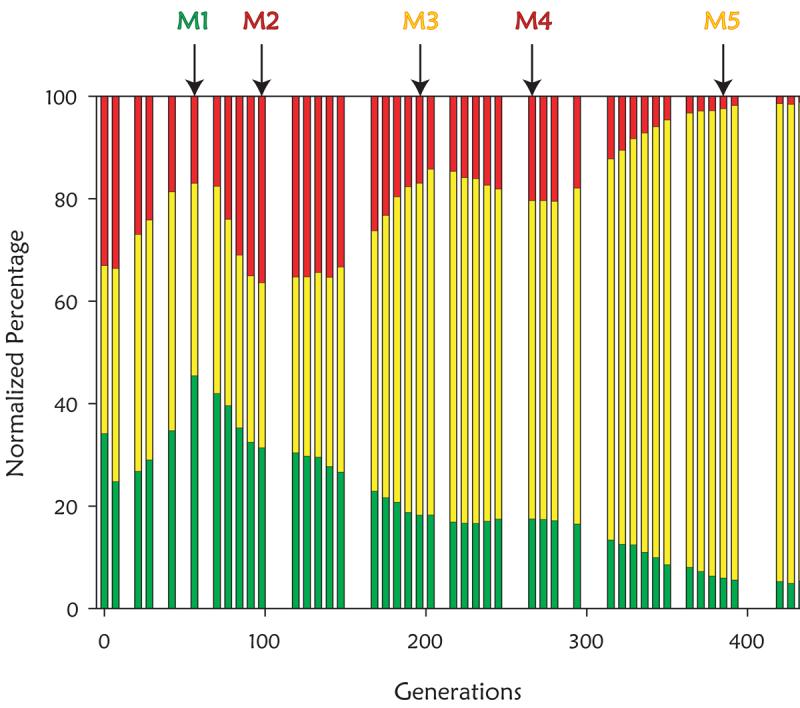
We followed this study by comprehensively identifying the mutations in 5
selected clones from the above population (M1 through M5), and showed that
mutations that increased glucose transport and signaling through the Ras
pathway were beneficial. Remarkably, we found that when two of the
beneficial mutations that increased glucose transport were combined (loss
of function mutations in MTH1 and amplification of
the HXT6/7 locus) that the fitness of the double mutant was less
than not only both single mutants, but less even than wild-type (Kvitek
and Sherlock, 2011). This is a clear case of reciprocal sign epistasis,
where mutations are mutually exclusive.
It was however clear to us, that while we had comprehensively analyzed 5
clones, that it was likely that we had only scratched the surface of the
complexity of the population dynamics, as well as the spectrum of
beneficial mutations. To address this, we performed high coverage
population sequencing with overlapping forward and reverse reads for error
correction. This allowed us to identify alleles in the population at
frequencies as low as 1%. Genotyping individuals from the populations for
the alleles abover 10% allowed us to determine the dynamics of the
evolutions as they progressed, revealing a much richer picture than was
possible just using 3 colors (Kvitek and Sherlock, 2013). One of the
three populations that we sequenced is shown below:
 References
References
-
Kao, K.C. and Sherlock, G. (2008). Molecular characterization
of clonal interference during adaptive evolution in asexual
populations of Saccharomyces cerevisiae. Nature
Genetics 40, 1499 - 1504.

-
Kvitek, D.J. and Sherlock, G. (2011). Reciprocal Sign Epistasis
between Frequently Experimentally Evolved Adaptive Mutations Causes a
Rugged Fitness Landscape. PLoS Genetics 7(4): e1002056.


-
Kvitek, D.J., Sherlock, G. (2013). Whole Genome, Whole Population
Sequencing Reveals That Loss of Signaling Networks Is the Major Adaptive
Strategy in a Constant Environment. PLoS Genetics 9(11):
e1003972.


|

