RESEARCH FOCUS
CARL has a mission to fuse bioengineering, data science and detailed physiological measurements in patients to develop novel management strategies. CARL focuses on the global complex arrhythmia pandemics of Atrial Fibrillation (AF), ventricular fibrillation (VF) and tachycardia (VT). Each is a significant cause of mortality and morbidity, and therapy for each remains suboptimal.
Atrial fibrillation (AF)
AF is the most common arrhythmia. While ablation is the most successful therapy, the success pulmonary vein isolation (PVI) is 50-70% at 1 year.
CARL is pioneering approaches to better understand which patients (‘phenotypes’) will respond to different therapies, fusing machine learning, models and natural language processing with clinical, imaging and electrical data.
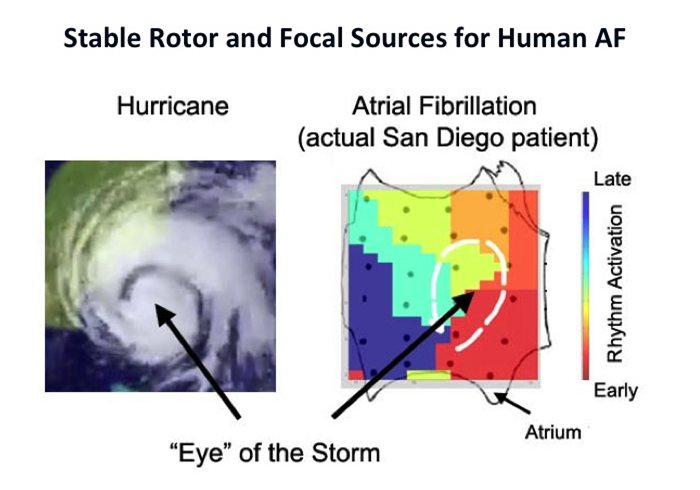
For patients at ablation, CARL pioneered computational approaches to map AF to identify ablation targets beyond PVI. Such localized drivers may represent focal or rotational activity, conduction slowing, fibrosis or autonomics. See Publications for more details
.
Ventricular fibrillation (VF)
VF is a major cause of sudden death, that affects over 250,000 individuals in the U.S. alone. Clinical VF is a relatively unstudied rhythm, even patients treated with ICDs may have recurrent collapse (‘syncope’) or shocks, with only early strategies for ablation. CARL has studied fundamental mechanisms of VF since 1996 – including repolarization and conduction abnormalities in the ventricles and conduction patterns – and has pioneered VF mapping to guide ablation from 2013 to the current time. See our ongoing NIH studies and Publications for more details.

Fundamental Cardiac Electrophysiology Studies in Human Hearts
Dynamic triggers such as ectopic beats, bursts of atrial flutter or varying autonomic balance may initiate AF, VF and VT despite fixed atrial architecture, surface curvature and fibrosis.
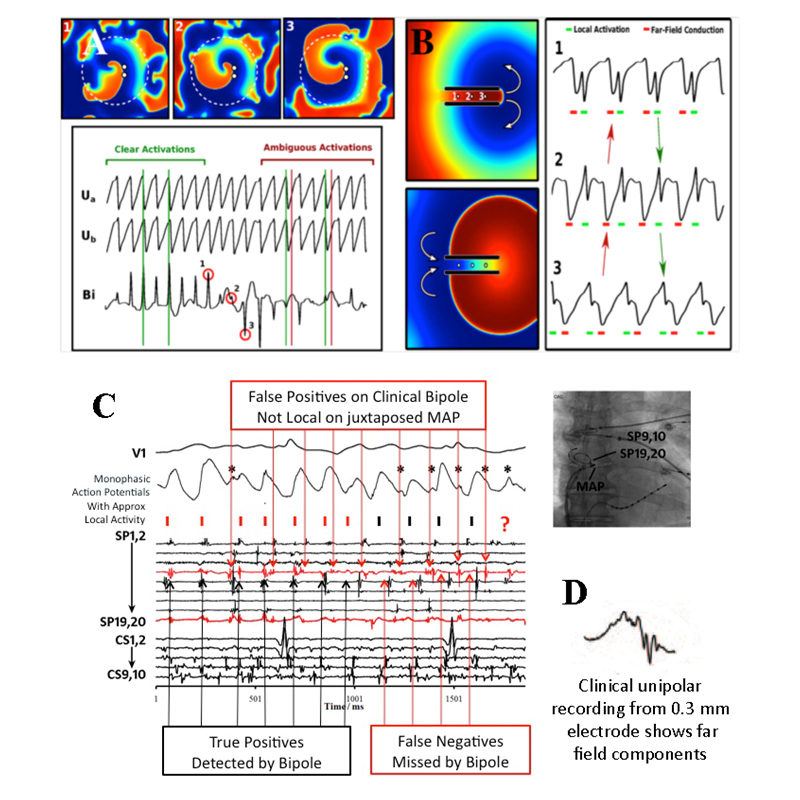
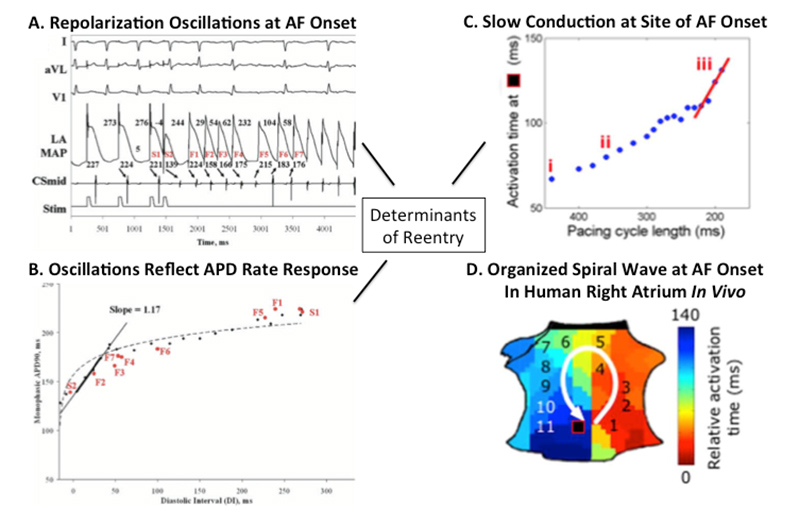
Since 1996, we have studied the dynamics of repolarization and conduction in human atria and ventricles, and why some triggers cause AF/VF while others do not. For AF, the figure shows how a single ectopic yields dramatic oscillations of left atrial action potential duration (APD) – if APD substantially shortens after early beats and lengthens after pauses (steep restitution). Figure 1C shows that triggers also slow conduction imminently before AF onset, producing spiral-wave re-entry at AF onset (figure 1D).
Mapping electrograms in complex arrhythmias is difficult and controversial, and a major unmet need. A growing gold standard is optical mapping of voltage-sensitive dye fluoresence, yet this cannot be applied to living patients due to dye toxicity and inability to visualize through blood. Figure 2A shows electrograms of AF in a computer model at a stable spiral wave (potential source) and surrounding ‘chaotic’ activity. We have reported in humans AF that bipolar signals often miss local activation yet detect unrelated far-field activations(figure 2C). In fig. 2D, electrograms may be more complex in a fibrotic milieu, that may further impact analysis.